|
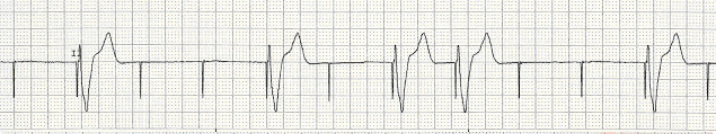
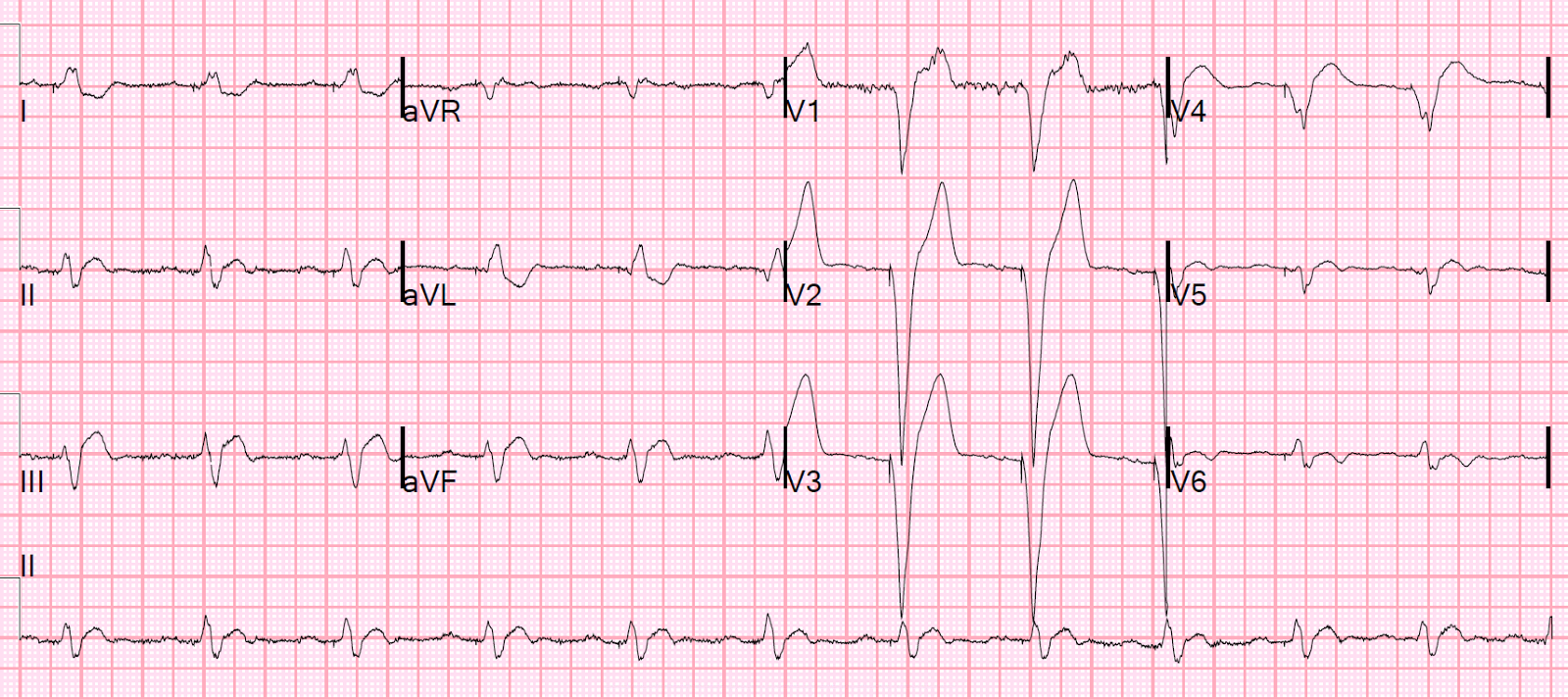
Algorithm to determine manufacturer using X-ray ( Jacob 2011)ĬaRDIA-X Algorithm Effect of Magnet on Pacemaker ( Core EM Video Link).The majority of the time, this information can be determined from the medical record or, the patient will carry a device manufacturer card with all of the information.In order to interrogate a pacemaker, the device manufacturer must be known.Treatment: replacement of single chamber pacemaker to dual chamber pacemaker.Symptoms: Syncope/Near-syncope, orthostatic dizziness, exercise intolerance, generalized weakness, palpitations.Occurs when there is intact sinus node function but atrium contracts with tricuspid/mitral valves closed (i.e.Loss of AV synchrony and loss of atrial “kick”.Pain, swelling, venous engorgement of the ipsilateral arm.Occur in only 0.5 -3.5% of patients due to collateralization.Common complication (30-50% with some venous obstruction) ( Kucher 2011).Cardiology consultation for removal and replacement.Needle aspiration: should only be performed under fluoroscopy to avoid cutting components of the device.Blood cultures: (+) in 20-25% of patients (commonly S.Signs/Symptoms: warmth, swelling, erythema, pain, fever.It may reflect adequate native conduction. Absence of paced complexes in a patient with a pacemaker does not always indicate pacemaker dysfunction.Atrial spike conducted through AV node and generates QRS.Conducted from intrinsic atrial activity.Second spike results in ventricular depolarization 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
